🩺 By Dr. Kulmeet Kundlas, MD — Board-Certified Internal Medicine, Shield Medical Group
Stromectol (ivermectin) is being studied for cancer treatment through Florida's $140M research fund. Learn what is ivermectin, its mechanisms, and potential outcomes.
Stromectol in Cancer Research: Florida's $140 Million Initiative Explained
Stromectol — the brand name for ivermectin — is making headlines again, but this time the conversation has nothing to do with COVID-19. The State of Florida has launched a massive $140 million research initiative to explore whether this Nobel Prize-winning antiparasitic drug can be repurposed forcancer treatment. Spearheaded by Governor Ron DeSantis and Florida First Lady Casey DeSantis, a breast cancer survivor herself, the Florida Cancer Innovation Fund represents one of the most ambitious state-funded drug repurposing efforts in recent history.
Watch: Can Ivermectin Really Help Fight Cancer? on YouTube
Below, we break down what is ivermectin, why researchers believe it may have anticancer properties, the specific mechanisms under investigation, and what patients should know moving forward. This educational overview is based on publicly available research discussions and clinical commentary — it isnotmedical advice or a recommendation for off-label use.
Watch the full medical video animated explainer from Kundlas MD, covering every aspect of this developing story.

What Is Stromectol? A Drug in Brief
Stromectol is the FDA-approved brand name forivermectin, a medication that has been in clinical use for over 50 years. Originally developed to treat parasitic infections — most notably river blindness (onchocerciasis) and resistant scabies — ivermectin earned its discoverers theNobel Prize in Physiology or Medicine in 2015.
Ivermectin works by targeting chloride channels inside parasite cells. This action causes paralysis of the parasite and eventually kills it. The drug is available in several forms:
- Ivermectin oral tablets— the most common prescription form for systemic parasitic infections
- Ivermectin cream— used topically, often prescribed for rosacea
- Ivermectin oral liquid— used in veterinary and some human applications
Whether you take ivermectin with food or on an empty stomach depends on the prescribing context. For parasitic infections, current guidelines generally recommend taking ivermectin with food to improve absorption, though your physician will provide specific ivermectin dosage instructions based on your weight and condition.
The safety profile of Stromectol for its approved indications is well-established. The human brain is protected by the blood-brain barrier, which prevents the medication from reaching the central nervous system in standard therapeutic doses. This is a critical safety feature that has allowed billions of ivermectin doses to be administered worldwide over five decades.
For a deeper look at theinternal medicine perspectiveon drug repurposing, our clinical team at Shield Medical Group stays current on emerging research.
Why Stromectol Is Being Studied for Cancer Treatment
The Florida Cancer Innovation Fund was born from deeply personal circumstances. First Lady Casey DeSantis was diagnosed with triple-negative breast cancer — one of the most aggressive and difficult-to-treat forms of the disease. When facing a diagnosis like this, every potential treatment option deserves exploration.
Governor DeSantis and the First Lady assembled a dedicated scientific team and initially secured $60 million in funding. That figure has since grown to$140 million, making this one of the largest state-backed cancer research investments focused on drug repurposing.
But why Stromectol? Over five decades of clinical use, physicians and researchers collected observations about ivermectin's broader biological effects beyond its antiparasitic mechanism. Preclinical studies — conducted in laboratory settings — have suggested that ivermectin may interact with cancer cells in ways worth investigating. These observations formed the scientific basis for the initiative.
This is how science progresses. Rather than relying on anecdotes or incomplete information, the Florida initiative puts together the funding, the research team, and a structured protocol to determine the facts. As noted in the video, this approach "kills all the controversies" by replacing speculation with rigorous evidence.
The Personal Dimension of Research Funding
Many breakthrough medical research programs begin with personal motivation. Research grants often come from individuals with political will, financial resources, or personal experience with a disease. The DeSantis family's initiative follows a well-established pattern in medical history — personal adversity driving public investment that benefits everyone.
Three Cancer Mechanisms Under Investigation
The Florida Cancer Innovation Fund is exploring three specific mechanisms by which Stromectol might affect cancer cells. Understanding who does ivermectin work on at the cellular level is central to this research. Here is a drug in brief overview of each pathway:
1. Direct Cancer Cell Death (Autophagy)
The first mechanism under study is whether ivermectin can program cancer cells to die — a process known asdirect cell deathor apoptosis. This concept is related to autophagy, the body's natural process of clearing old, damaged, or senescent cells.
You may have heard of autophagy in the context of intermittent fasting. The same cellular recycling concept applies here. Researchers are investigating whether ivermectin can trigger cancer cells to self-destruct while leaving healthy cells intact. If confirmed, this could represent a powerful and relatively low-toxicity treatment approach.
2. Reducing Chemotherapy Resistance
One of the biggest challenges in oncology ischemotherapy resistance. Cancer cells exposed to chemotherapy can adapt and become resistant, rendering previously effective treatments useless.
The second mechanism under investigation is whether ivermectin can work alongside existing chemotherapy drugs to reduce or overcome this resistance. If Stromectol can make cancer cells more vulnerable to chemotherapy, it could dramatically improve outcomes for patients whose tumors have stopped responding to standard treatments.
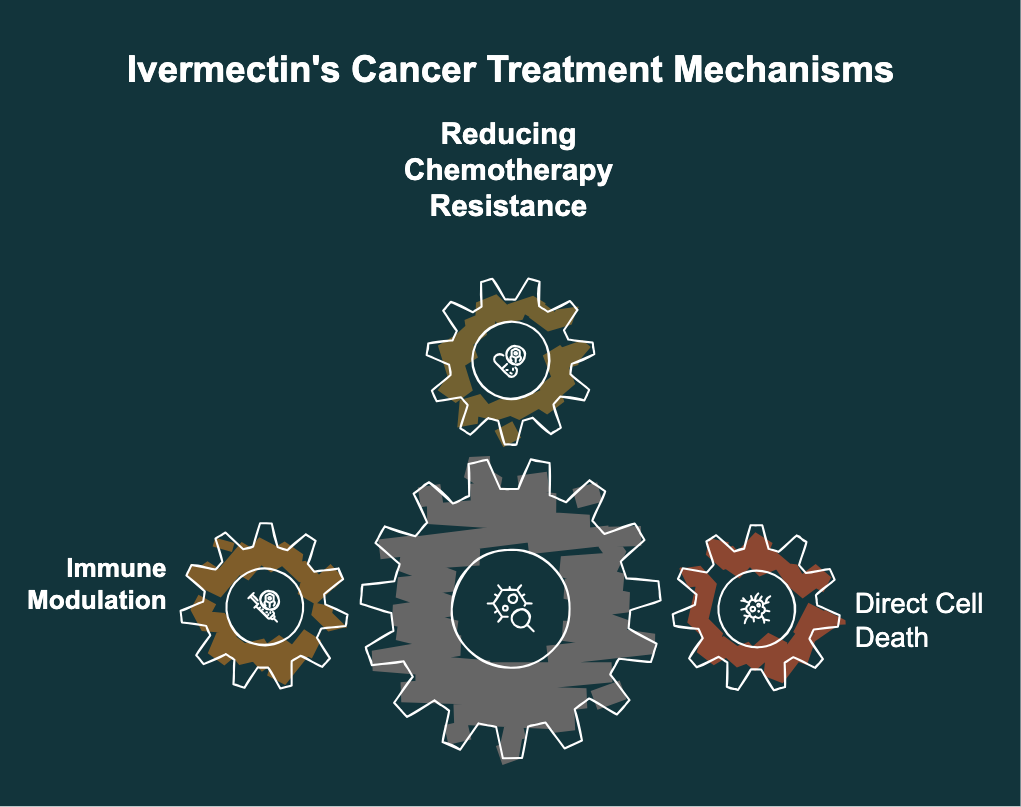
3. Immune Modulation — Making Cold Tumors Hot
The third and perhaps most exciting mechanism involvesimmune modulation. Cancer cells are remarkably smart. They can become metabolically "cold," meaning they essentially hide from the immune system and from chemotherapeutic drugs.
When cancer cells are metabolically "hot," treatments can reach them and destroy them. When they turn cold, they become invisible to treatment. Researchers postulate that ivermectin may function as anivermectin antiviral-like immune modulator — not antiviral in the traditional sense, but capable of reactivating the immune system's ability to recognize and attack cancer cells.
If ivermectin can make cold cancer cells hot again, it would be a significant breakthrough in making existing immunotherapies and chemotherapies more effective.
Ivermectin Side Effects and Safety at Higher Doses
One critical question the Florida research must address involvesivermectin side effectsat the higher doses proposed for cancer treatment. The doses being considered are significantly higher than those used for parasitic infections.
At standard therapeutic levels, ivermectin side effects are generally mild and may include:
- Dizziness or lightheadedness
- Nausea or mild gastrointestinal discomfort
- Skin rash or itching (particularly during parasite die-off)
- Muscle or joint pain
The blood-brain barrier provides significant protection at standard doses. However, at elevated doses, the safety margin narrows. Understanding how far the dosage can be pushed while maintaining an acceptable safety profile is one of the primary objectives of this research initiative.
Patients should be aware that ivermectin withdrawal from ongoing parasite treatment should always be managed under physician supervision. The interaction profile — including concerns about ivermectin with alcohol and ivermectin with mebendazole combinations — requires careful clinical oversight.
This is precisely why structured research like the Florida Cancer Innovation Fund exists: to determine safe dosage ranges through controlled studies rather than guesswork. For questions about medication interactions orour clinical approach, Shield Medical Group welcomes patient inquiries.
Lessons from COVID-19 and the Path Forward for Stromectol Research
Any discussion of Stromectol inevitably raises memories of the COVID-19 era. During the pandemic, ivermectin was widely discussed — and hotly debated — as a potential treatment for the virus. The reality, as clinical evidence ultimately showed, was that ivermectin was not effective against COVID-19.
However, as emphasized in the medical videos animated explanation above, the medical community's attempt to repurpose existing medications during an unprecedented crisis was understandable. When there were no approved treatments available and patients were dying, physicians explored every option. Some early observations were over-exaggerated out of desperation, not malice.
The Florida Cancer Innovation Fund represents a fundamentally different approach:
- Pre-established funding— $140 million secured before the research begins
- Dedicated scientific team— assembled with specific cancer research expertise
- Defined timelines and protocols— structured guidelines for investigation
- Specific mechanisms— targeted research on identified biological pathways
This is evidence-based science in action. If the research yields positive results, larger clinical trials will follow. If the results are negative, the investigation stops — and that clarity is equally valuable.

The Clinical Reality for Patients Today
Despite growing public interest, physicians are currently unable to prescribe ivermectin for cancer treatment. Doctors are held to evidence-based standards and cannot prescribe medications based on anecdotes or incomplete research. As shared in the video, patients have sought ivermectin prescriptions from oncologists and primary care physicians, only to be told that the evidence does not yet support this use.
Some patients have traveled to states where ivermectin for human use can be procured over the counter, and some report positive experiences. However, individual testimonials do not constitute clinical evidence. The Florida research initiative exists specifically to generate the rigorous data needed to make informed medical decisions.
Both osteopathic medicine and allopathic medical training emphasize this same principle: treatment decisions must be grounded in evidence. Whether you work in nurse education or direct patient care, this standard protects patients from harm.
If you have questions about emerging treatments or cancer support resources, pleasecontact Shield Medical Groupto schedule a consultation.
What Patients and Families Should Know
For patients and families following this story, here are the key takeaways:
- Stromectol (ivermectin) is being studied— not approved — for cancer treatment
- The research is well-funded ($140 million) and scientifically structured
- Three specific anticancer mechanisms are under investigation
- Donotself-medicate with ivermectin for cancer — ivermectin dosage for this purpose has not been established
- Talk to your oncologist or primary care physician about any interest in emerging treatments
- The results of this research — positive or negative — will advance our understanding
The medical community is watching the Florida Cancer Innovation Fund closely. If the data supports repurposing Stromectol for cancer treatment, it could open a new chapter for a medication that has already saved millions of lives worldwide.
Medical Disclaimer:This content is for educational purposes only and does not constitute medical advice, diagnosis, or treatment. Always consult your healthcare provider before starting or changing any medication. Ivermectin is not FDA-approved for cancer treatment. Content reviewed by Kulmeet Kundlas MD, Board-Certified Internal Medicine, Shield Medical Group, Sebring and Lake Wales, Florida.
Frequently Asked Questions
Frequently Asked Questions About Stromectol and Ivermectin
Is ivermectin an antibiotic?
No, ivermectin is not an antibiotic. It is an antiparasitic medication that works by targeting chloride channels in parasite cells, causing paralysis and death of the organism. It belongs to a class of drugs called avermectins and has no antibacterial activity.
Is ivermectin over the counter?
In the United States, ivermectin tablets for human use generally require a prescription from a licensed physician. Some states have varying regulations, and veterinary formulations (such as ivermectin paste or ivermectin at tractor supply stores) are available without prescription but arenotintended for human consumption and may contain dangerous inactive ingredients.
Is ivermectin an antiviral?
Ivermectin has shown some antiviral properties in laboratory (in vitro) studies, but clinical trials have not confirmed meaningful antiviral effectiveness in humans. The FDA has not approved ivermectin as an antiviral medication, and its use during COVID-19 ultimately did not demonstrate clinical benefit.
Is ivermectin safe for pregnant women?
Ivermectin is classified as a Pregnancy Category C medication, meaning animal studies have shown adverse effects on the fetus and there are no adequate human studies. Pregnant women should not take ivermectin unless the potential benefit clearly justifies the risk, and only under direct physician supervision.
What ivermectin is best for humans?
The FDA-approved form of ivermectin for humans is Stromectol, available as oral tablets in 3 mg strength. Only FDA-approved human formulations should be used — veterinary products such as ivermectin paste for horses or ivermectin for dogs contain different concentrations and inactive ingredients that can be harmful to people.
Is ivermectin safe for dogs?
Ivermectin is used in veterinary medicine to treat heartworm and certain parasites in dogs. However, some dog breeds — particularly collies and related herding breeds — carry a genetic mutation (MDR1) that makes them extremely sensitive to ivermectin. Dog owners should always consult a veterinarian before administering ivermectin to pets.
Is ivermectin an antifungal?
No, ivermectin is not an antifungal medication. It is specifically designed to target parasitic organisms and has no established activity against fungal infections. Antifungal treatment requires entirely different classes of medication.